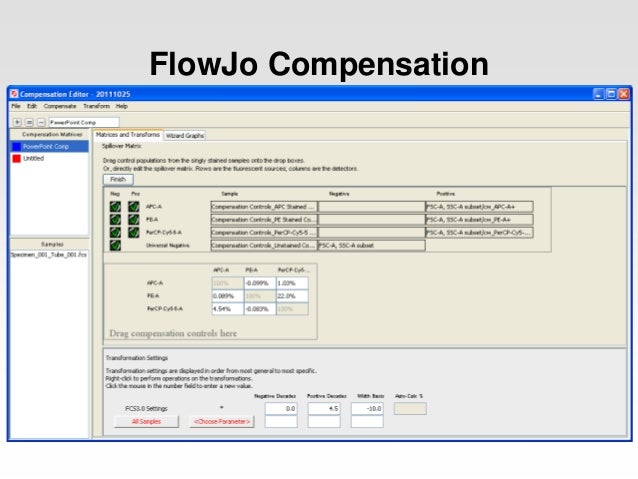
 The autofluorescence of the positive population, before staining, should be the same as the negative control. The positive should be at least as bright as anything that will be encountered in the experiment and should form at least 10% of the population. These controls should be solely used to set compensation. Compensation controls are required for each fluorochrome and should contain both a positive and a negative population. Some fluorochrome combinations should be avoided if possible (eg APC and PE-Cy5), given the high degree of emission overlap.Dead cells, clumps and debris should be excluded from further analysis. Adjust forward scatter and side scatter so that the cell population is clearly delineated. Set voltages for fluorescence channels using an unstained sample.Ensure that the cytometer is performing within specifications using standard beads.We always recommend reviewing the flow cytometer manufacturer's instructions for detailed compensation guidelines. However, the following guidelines should be suitable in most cases. The procedure for setting correct fluorescence compensation is essentially the same on any cytometer but there are differences between the various available instruments, which makes it difficult to provide a "one size fits all" protocol. (PE-Cy5 + PE overlap) - (PE overlap) = accurate PE-Cy5 results General procedure Adjust the compensation settings until no PE signal is seen in the PE-Cy5 channel (see the procedure below). 
Observe the signal in both PE and PE-Cy5 channels.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
